
In addition to approving funding for breast cancer related brain metastases last week, the CIRM Board also approved an additional $19.7 million geared towards our translational research program. The goal of this program is to help promising projects complete the testing needed to begin talking to the US Food and Drug Administration (FDA) about holding a clinical trial.
Before getting into the details of each project, here is a table with a brief synopsis of the awards:
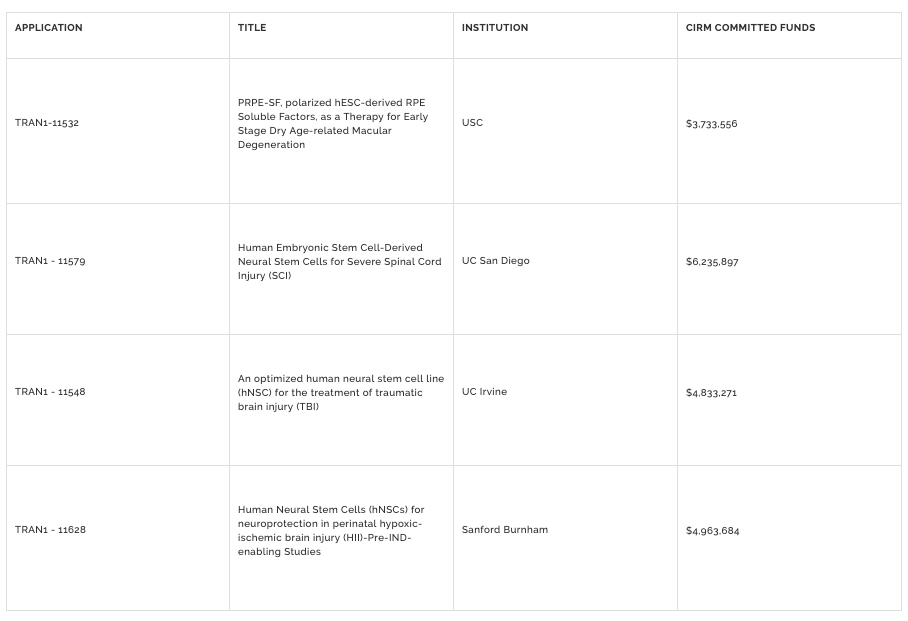
TRAN1 – 11532
$3.73 million was awarded to Dr. Mark Humayun at USC to develop a novel therapeutic product capable of slowing the progression of age-related macular degeneration (AMD).
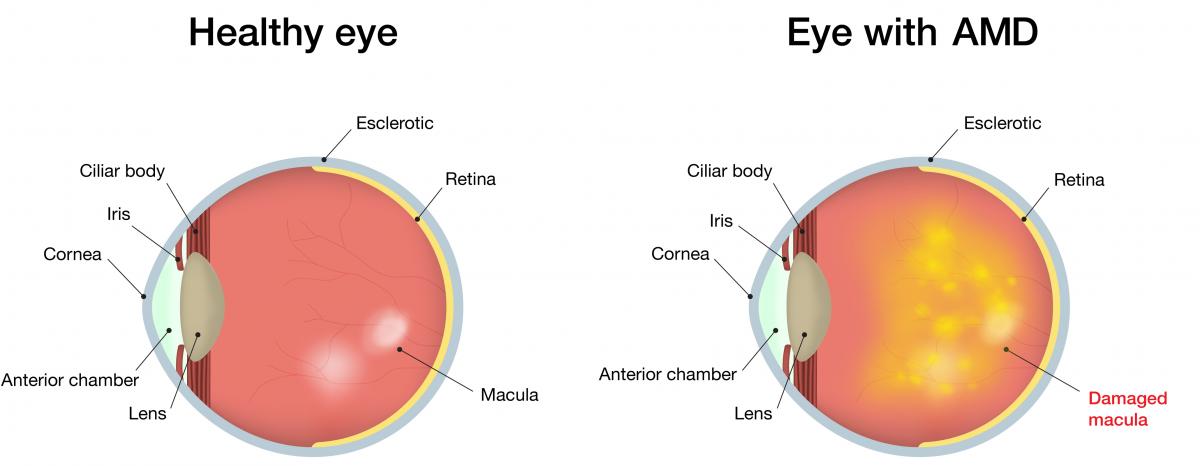
AMD is an eye disease that causes severe vision impairment, resulting in the inability to read, drive, recognize faces, and blindness if left untreated. It is the leading cause of vision loss in the U.S. and currently affects over 2 million Americans. By the year 2050, it is projected that the number of affected individuals will more than double to over 5 million. A layer of cells in the back of the eye called the retinal pigment epithelium (RPE) provide support to photoreceptors (PRs), specialized cells that play an important role in our ability to process images. The dysfunction and/or loss of RPE cells plays a critical role in the loss of PRs and hence the vision problems observed in AMD. One form of AMD is known as dry AMD (dAMD) and accounts for about 90% of all AMD cases.
The approach that Dr. Humayun is developing will use a biologic product produced by human embryonic stem cells (hESCs). This material will be injected into the eye of patients with early development of dAMD, supporting the survival of photoreceptors in the affected retina.
TRAN1 – 11579
$6.23 million was awarded to Dr. Mark Tuszynski at UCSD to develop a neural stem cell therapy for spinal cord injury (SCI).
According to data from the National Spinal Cord Injury Statistical Center, as of 2018, SCI affects an estimated 288,000 people in the United States alone, with about 17,700 new cases each year. There are currently no effective therapies for SCI. Many people suffer SCI in early adulthood, leading to life-long disability and suffering, extensive treatment needs and extremely high lifetime costs of health care.
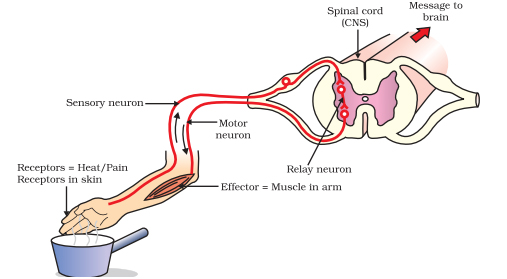
The approach that Dr. Tuszynski is developing will use hESCs to create neural stem cells (NSCs). These newly created NSCs would then be grafted at the site of injury of those with SCI. In preclinical studies, the NSCs have been shown to support the formation of neuronal relays at the site of SCI. The neuronal relays allow the sensory neurons in the brain to communicate with the motor neurons in the spinal cord to re-establish muscle control and movement.
TRAN1 – 11548

$4.83 million was awarded to Dr. Brian Cummings at UC Irvine to develop a neural stem cell therapy for traumatic brain injury (TBI).
TBI is caused by a bump, blow, or jolt to the head that disrupts the normal function of the brain, resulting in emotional, mental, movement, and memory problems. There are 1.7 million people in the United States experiencing a TBI that leads to hospitalization each year. Since there are no effective treatments, TBI is one of the most critical unmet medical needs based on the total number of those affected and on a cost basis.
The approach that Dr. Cummings is developing will also use hESCs to create NSCs. These newly created NSCs would be integrated with injured tissue in patients and have the ability to turn into the three main cell types in the brain; neurons, astrocytes, and oligodendrocytes. This would allow for TBI patients to potentially see improvements in issues related to memory, movement, and anxiety, increasing independence and lessening patient care needs.
TRAN1 – 11628
$4.96 million was awarded to Dr. Evan Snyder at Sanford Burnham Prebys to develop a neural stem cell therapy for perinatal hypoxic-ischemic brain injury (HII).
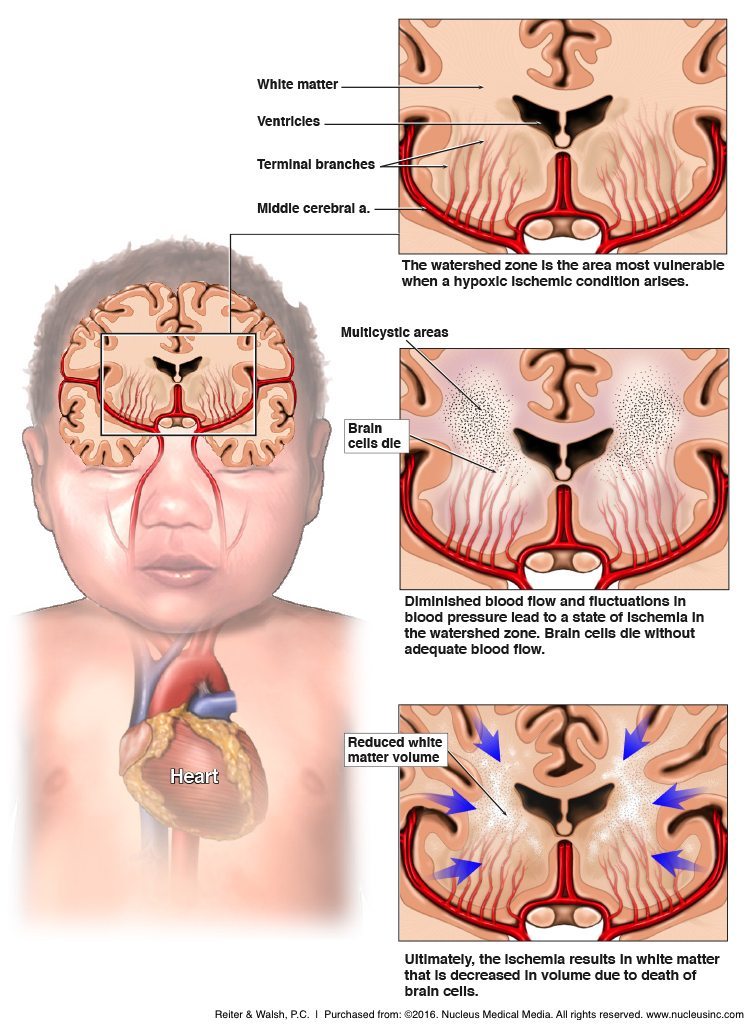
HII occurs when there is a lack of oxygen flow to the brain. A newborn infant’s body can compensate for brief periods of depleted oxygen, but if this lasts too long, brain tissue is destroyed, which can cause many issues such as developmental delay and motor impairment. Current treatment for this condition is whole-body hypothermia (HT), which consists of significantly reducing body temperature to interrupt brain injury. However, this is not very effective in severe cases of HII.
The approach that Dr. Snyder is developing will use an established neural stem cell (NSC) line. These NSCs would be injected and potentially used alongside HT treatment to increase protection from brain injury.
my son had spinal court injury in 4/4/2019 i need help search to stem cells treatment for him
Dear Alfred, the only clinical trial that might be applicable and appropriate for your son is this one.
https://clinicaltrials.gov/ct2/show/NCT01772810?term=stem+cells&recrs=a&cond=Spinal+Cord+Injuries&cntry=US&rank=2
I hope that helps.