THIS BLOG IS ALSO AVAILABLE AS AN AUDIO CAST
One of the huge advantages of a stem cell agency like CIRM (not that there is anything out there quite like us, but anyway) is our ability to support projects as they progress from a great idea to a therapy actually being tested in people.
Exhibit A on that front came via a news release from ViaCyte, a company that is developing a new approach to helping people with severe Type 1 Diabetes (T1D).
Unlike type 2 diabetes, which is largely diet & lifestyle related and develops over time, T1D is an autoimmune condition where the person’s immune system attacks and destroys the insulin-producing cells in the pancreas. Without those cells and insulin the body is not able to regulate blood sugar levels and that can lead to damage to the heart, kidneys, eyes and nerves. In severe cases it can be fatal.
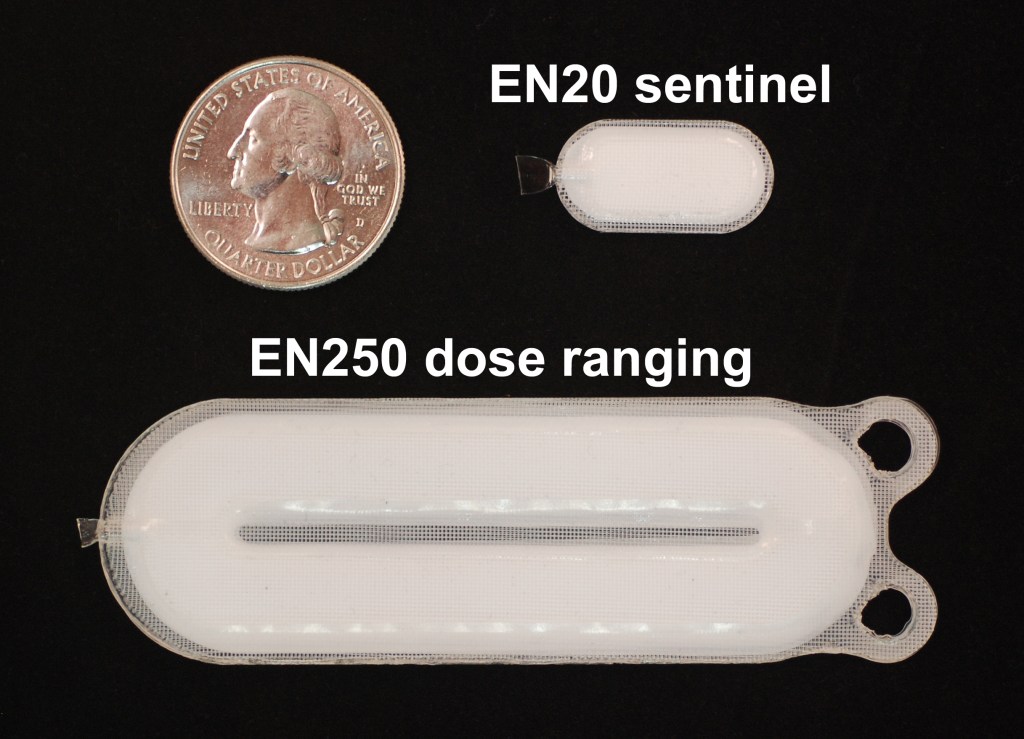
ViaCyte (which has been supported with more than $72 million from CIRM) has developed a pouch that can be implanted under the skin in the back. This pouch contains stem cells that over a period of a few months turn into insulin-producing pancreatic islet cells, the kind destroyed by T1D. The goal is for these cells to monitor blood flow and when they detect blood sugar or glucose levels are high, can secrete insulin to restore them to a safe level.
They tested this approach in 15 patients in a Phase 1 clinical trial in Canada. Their findings, published in the journals Cell Stem Cell and Cell Reports Medicine, show that six months after implantation, the cells had turned into insulin-producing islet cells. They also showed a rise in C-peptide levels after patients ate a meal. C-peptides are a sign your body is producing insulin so the rise in that number was a good indication the implanted cells were boosting insulin production.
As Dr. James Shapiro, the Chair of Canada Research and one of the lead authors of the study says, that’s no small achievement: “The data from these papers represent a significant scientific advance. It is the first reported evidence that differentiated stem cells implanted in patients can generate meal-regulated insulin secretion, offering real hope for the incredible potential of this treatment.”
And that wasn’t all. The researchers say that patients spent 13 percent more time in the target range for blood sugar levels than before the treatment, and some were even able to reduce the amount of insulin they injected.
Now this is only a Phase 1 clinical trial so the goal was to test the safety of the pouch, called PEC-Direct (VC-02), to see if the body would tolerate it being implanted and to see if it is effective. The beauty of this method is that the device is implanted under the skin so it can be removed easily if any problems emerge. So far none have.
Ultimately the hope is that this approach will help patients with T1D better regulate their blood sugar levels, improve their health outcomes, and one day even achieve independence from the burden of daily insulin injections.
Stem cells are great advantage for treatment of many diseases. In many adult tissues, stem cells are ease to be isolated, cultures and availability in many tissues. Their immunomodulatory properties and lack of ethical issues giving hope for both autologous and allogenic transplantation. However, stem cell -based therapy may produce undesirable effects. Many cumulative factors can give a response in the form of spontaneous tumor transformation, such as, 1)The ability of stem cells to proliferate for long periods of time, high viability and resistance to apoptosis can easily develop tumorigenesis. 2)Donor’s age, host tissue, growth regulators expressed by recipient tissues and mechanism that control behavior of stem cells at the target site. 3)Manipulation and longterm in vitro cultures of stem cells can cause genetic instability and chromosome aberration
Current clinical trial by using PSC-derived pancreatic progenitors which were fully encapsulated device and implanted into patients with T1D. The observation revealed that implanted device acquired neovasculature and PEC-01 cells matured into insulin-expressing β cells in T1D patients. The results showed that 63% of trial patients had stimulated C-peptide response which was indicator to determine endogenous β cell function. The unequivalent response of T1D patients to implanted PSC-derived cells indicates that pancreatic progenitors require growth factors to growth and differentiation. The different ability of T1D patients produce growth factors and support maturation of progenitors causing diversity of findings. During the process of maturation, stem cells require multiple stages of growth and differentiation to produce mature and functioning cells. Each stage of maturation requires specific growth factor and timing for development. Hence, T1D patients require additional supply of specific growth factor to support progenitors development on certain period of time. As all trial patients are undergoing immunosuppressive treatment, the possibility of patients develop cancers for longterm period of time is unavoidable. Of course, those aberrant cells are unable to be removed by autoimmune response. Thus, extensive follow up is essential to prevent the patients from developing cancers. The short period of observation within 1-2 years does not support the safety and efficacy of implanted PSC-derived cells in T1D patients. The longterm follow up of trial patients are essential to protect the health and well being of T1D patients.