As we enter the new year, CIRM’s 2017 Annual Report will be posted in a few short weeks! Here’s a sneak peek at CIRM’s progress in clinical trials.

2017 CIRM Annual Report
At the start of 2017, we set a goal of finding and funding 12 new, high quality clinical trials. We easily beat that goal, funding 16, in a wide variety of conditions from ALS (also known as Lou Gehrig’s disease) to cancer and diabetes. That means we have now funded a total of 43 different projects in clinical trials and enrolled more than 700 people in those trials.
Here’s a look at the different kinds of stem cells and diseases are involved in those clinical trials:
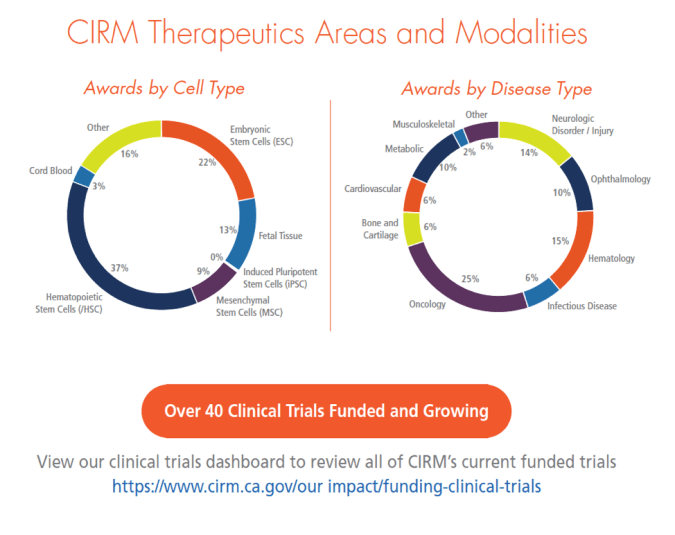
Funding those 16 new clinical trials means we have now funded 26 new trials in the last two years, putting us ahead of schedule to meeting our goal of 50 new clinical trials by 2020.
When we fund clinical programs, we truly partner with these programs and give them support – financially, operationally and strategically.
CIRM assists investigators in the application process so they can best articulate their research proposal in a way that can be optimally evaluated by our independent peer review group for funding. By putting applications through a rigorous review process, we select programs with the highest probability of success. You will hear from one of our GWG members, the external panel that reviews our grants for funding, in the Annual Report.
CIRM provides funding at a critical stage when programs are not yet able to get sufficient funding because they are felt to be “too early” or “too risky” for traditional investors. By funding these investigators to conduct important early work, CIRM “de-risks” the projects, and we have already seen how this has allowed “high risk but high reward” programs to attract investors and commercialization partners. We will feature examples of these follow-on investments in the Annual Report.
In addition to funding clinical trials, CIRM brings in critical expertise and resources for these programs. Clinical Advisory Panels (CAPs), composed of CIRM science officers, external experts and patient representatives, meet on a quarterly basis for each program to help them overcome obstacles and meet project milestones. CIRM has created the Stem Cell Center – a stem cell-specific research organization that helps investigators navigate the best regulatory pathways, provides access manufacturing resources, operational clinical trial support and strategic resources for delivering successful products to patients.
In short, we do everything we can to try and ensure those clinical trials have the best possible chance to be successful.
With a growing number of clinical trials to track, and more on the way, we needed a new tool to make it easier to see, at a glance, the trials we are funding, and all the key details of each program.
So, we created the Clinical Trials Dashboard to let you sort each trial by disease type, researcher, company or institution, and phase, as well as how many patients are to be enrolled. It also includes links to the www.clinicaltrials.gov website – a list of clinical trials registered with the National Institutes of Health – with details about patient eligibility and how to apply to be part of the trial.
The Dashboard is our way of making it as easy as possible for you to find the information you need, when you need it.
On Thursday, we’ll introduce you to one of the patients involved in a CIRM-funded clinical trial for cancer.
Thanks for the great news!