Meet another one of our talented CIRM Scholars, Helen Fong. She is currently a Research Scientist at the Gladstone Institutes and did her graduate work at the University of California, Irvine. Her passions include stem cells, disease modeling, and playing with differentiation protocols – the processes that tell stem cells to mature into specific tissues. As a CIRM Scholar, part of our educational training programs, Helen published four articles where she was listed as the first author. Her most recent one was a stellar study published in Stem Cell Reports using induced pluripotent stem cells (iPSCs) to model and understand a nerve cell-destroying brain disease called frontotemporal dementia.
We interviewed Helen to learn more about her work in stem cell research.
Q: What was your graduate school research on?
HF: I did my graduate work in the lab of Dr. Peter Donovan, who is a prominent germ cell and stem cell scientist, and was newly recruited to UCI when I began my studies. I was his first graduate student from UCI. Dr. Donovan’s research was focused on understanding the regulation of early human development using embryonic stem cells (ESCs) and how to improve human pluripotent stem cell culture. He was also interested in understanding the biological mechanisms that keep stem cells pluripotent (the ability to become all the other cell types in the body) and the genetic factors that are important for maintaining pluripotency. My graduate research was on understanding the basic biology of human ESCs. Specifically, I studied the role of the gene Sox2 in maintaining stem cell pluripotency and self renewal in human ESCs.
Q: What about your postdoctoral research?
HF: After my PhD, I decided to continue to work with stem cells because I knew that the field would continue to grow. There was still so much to be learned about these unique cells. I also genuinely enjoyed working with stem cells and couldn’t imagine not seeing them every day. I realized that I had a solid understanding of the basic biology of ESCs, but I wanted to use stem cells to study human disease. This ability is one of the huge selling points of working with human induced pluripotent stem cells (iPSCs) [which are created by reprogramming adult cells back to a pluripotent state]. The Gladstone Institutes was an excellent place to continue my training and to begin using iPSCs to understand neurological disease. I joined Dr. Yadong Huang’s lab in 2011 and am currently using human iPSCs to study brain degenerative diseases including frontotemporal dementia (FTD), progressive supranuclear palsy (PSP), and Alzheimer’s disease (AD).
My recent publication in Stem Cell Reports used human iPSCs from a patient with FTD as a model to understand the mechanisms behind this condition. This patient carried a rare genetic mutation in the MAPT gene called TAU-A152T. Several studies have reported a number of patients with this specific mutation that could put them at risk for developing FTD, PSP, and AD. However, it wasn’t clear what this mutation was doing to cause these disorders.
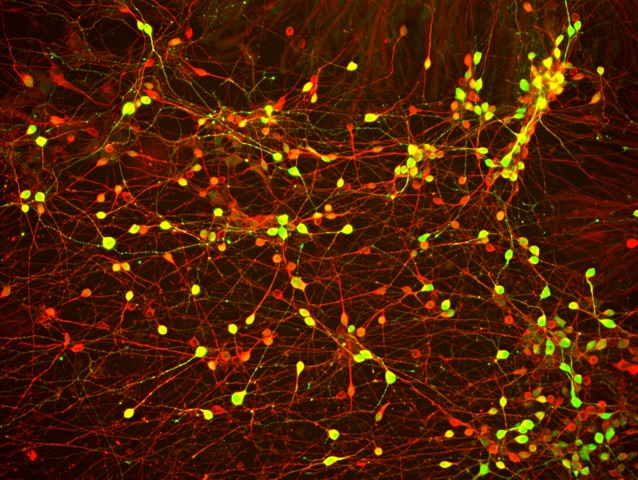
One of the ways you can study neurodegenerative diseases is using stem cells derived from patients harboring the disease causing mutations. We obtained human iPSCs made from the skin cells of a patient with FTD and this TAU mutation. I then used zinc finger nuclease (ZFN) genome editing technology to genetically correct the mutation back to the wild type (normal) sequence to see if removing this mutation in the patient iPSCs would generate healthier neurons (nerve cells) that don’t have symptoms of FTD. I was able to study the disease-causing effects of the TAU mutation by comparing healthy neurons I made from the corrected (normal) iPSC line to diseased neurons made from the TAU mutant iPSC line.
The neurons that I differentiated from the iPSCs carrying the TAU mutation showed an increase in TAU protein fragmentation [meaning the protein gets degraded and isn’t present in its normal form], an abnormal characteristic that can be associated with FTD and AD. We didn’t see this phenomenon in the neurons from the corrected (normal) human iPSCs, indicating that removal of this TAU mutation could improve the symptoms of these diseases. These results were exciting because we now had a culprit for what could be causing disease in these patients with this mutation. There is still much to be learned about the mechanisms of this mutation and the iPSCs have been an invaluable resource.
Q: What was your experience like as a CIRM scholar?
HF: CIRM has funded me for almost all of my stem cell training and research. I got my first CIRM training grant as a graduate student at UCI in 2006 and was funded for three years as a postdoc at the Gladstone. So I have CIRM to thank for all of my training.
When I first started out as a CIRM scholar, I believe I was part of one of their earlier pre-doctoral training grant programs. As the program expanded, I got to meet many of the other trainees at CIRM research conferences and interact with prominent stem cell scientists in the area. This was an incredible experience because I was exposed to stem cell research outside of my own institute, and I was able to meet all the big players in the field!
CIRM has also been very generous and provided me a travel allowance to attend any scientific conference of my choice. Over the years, I’ve gone to a lot of conferences nationally and internationally including ISSCR (International Society for Stem Cell Research), Keystone symposia, and the Society for Neuroscience (SfN). I have given scientific talks both at Keystone and SfN, and they proved to be excellent exposure for my work as well as a good place to get feedback. Another one of my favorite perks was the ability to purchase reagents for my own work at my own discretion, which gave me some freedom in dictating which direction I wanted my project to go. If I wanted to study a particular protein and needed a specific antibody to do that, I was able to get it with my CIRM funding.
Q: What’s next for your career?
HF: Currently, I am hoping to wrap up the project I am working on in the lab right now and generate a publication. I plan to continue to work on stem cells in the next step of my career and to work on challenging and cutting-edge projects. I feel fortunate for all the training and resources that I’ve received that got me to where I am today, and I hope to pass on many of my skills and knowledge to budding, young scientists.
Q: What is your favorite thing about being a scientist?
HF: I really enjoy the fact that I have so much control over the fate of my stem cells. They have the ability to turn into almost any cell type, and we’ve developed so many protocols to guide them into the exact cell type we want. They don’t always behave, but I think figuring out the personality of each and every cell line is part of the fun.
Related Links:

Is there any type of treatment for Degenerative Arthritis???