
Getting older is a normal, at times existential, part of life. The outward changes are abundant and noticeable: thinning of the hair, greying of the hair, and added lines to the face. There are also changes that happen that are not so abundantly clear in terms of outward appearance: slowing of healing time for bone fractures and a gradual loss of bodily function. The process of aging poses one very fundamental question — Could understanding how stem cells age lead to a greater understanding of how diseases develop? More importantly, could it guide the approach towards developing potential treatments? Two different studies highlight the importance of evaluating and understanding the process of aging in stem cells.
The first study, led by Dr. Michael Fehlings, looked at the impact of donor age in relation to stem cell therapies for spinal cord injuries (SCI). Dr. Fehlings, with a team of investigators from the University of Toronto and Krembil Research Institute, University Health Network, used an adult rat model to look at how cells derived from young vs. old stem cells affected tissue regeneration and recovery after a spinal cord injury.
Some rats with a SCI received cells derived from stem cells in the umbilical cord blood, which are considered “young” stem cells. The other rats with a SCI received cells derived from stem cells in the bone marrow, which are considered “old” stem cells. The results showed, ten weeks after treatment, that rats given the “young” stem cells exhibited a better recovery in comparison to those given the “old” stem cells.
In a press release, Dr. Fehlings stated that,
“Together, this minimally invasive and effective approach to cell therapy has significant implications on the treatment of traumatic cervical SCI and other central nervous system injuries. These results can help to optimize cell treatment strategies for eventual use in humans.”
The full results to this study were published in Stem Cells Translational Medicine.
The second, separate study, conducted by Dr. Stephen Crocker at UConn Health, looks at brain stem cells in people with multiple sclerosis (MS), a neurodegenerative disease caused by the inflammation and destruction of the insulation around the nerves, also known as myelin. Problems with insulation around the nerves can prevent or complicate the electrical signals sent from the brain to the body, which can lead to problems with walking or other bodily movements.
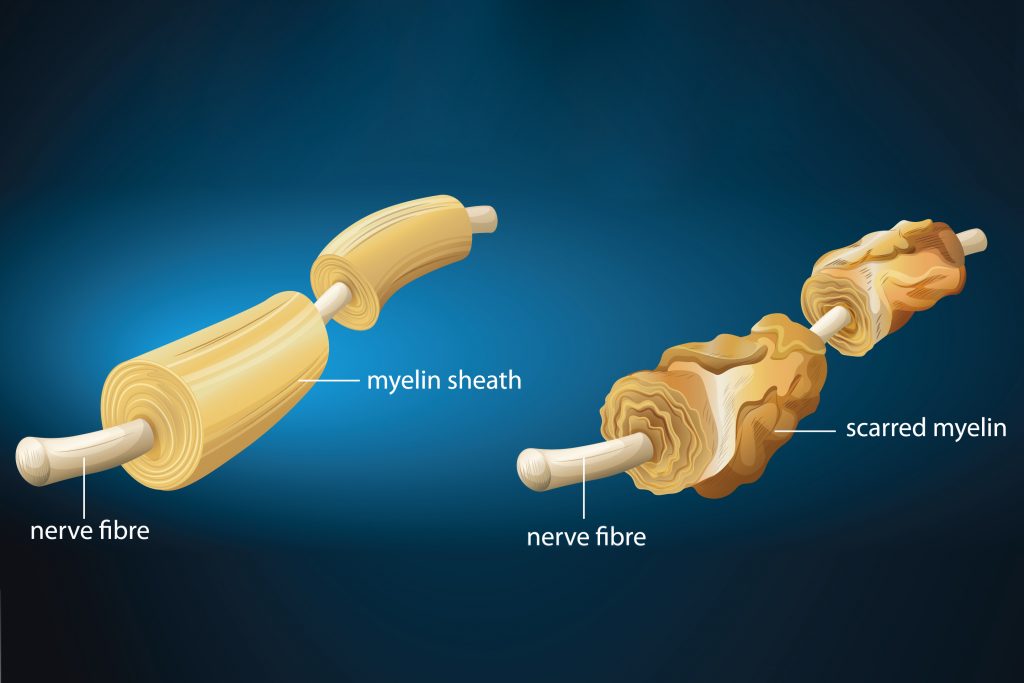
Dr. Crocker and his team found that brain stem cells in patients with MS look much older when compared to the brain stem cells of a healthy person around the same age. Not only did these brain stem cells look older, but they also acted much older in comparison to their healthy counterparts. It was also discovered that the brain stem cells of MS patients were producing a protein that prevented the development of insulation around the nerves. What is more remarkable is that Dr. Crocker and his team demonstrated that when this protein is blocked, the insulation around the nerves develops normally again.
In a press release, Dr. Valentina Fossati, a neurologist at the New York Stem Cell Foundation who evaluated these brain stem cells, stated that,
“We are excited that the study of human stem cells in a dish led to the discovery of a new disease mechanism that could be targeted in much-needed therapeutics for progressive MS patients.”
The complete study was published in the Proceedings of the National Academy of Sciences (PNAS).
ANYTHING ON ALS STEM CELL???
Here is a clinical trial for ALS that is underway right now. https://clinicaltrials.gov/ct2/show/NCT03280056